Testing for
AHP
COMMON TESTING OPTIONS
This testing information is provided for educational and reference purposes only. It is not intended to replace the independent medical judgment of any healthcare professional.
Consider ordering random (spot) urine tests to look for substantially elevated levels of aminolevulinic acid (ALA), porphobilinogen (PBG), and porphyrins, the neurotoxins that are believed to cause attacks and disease manifestations.1-5
RANDOM (SPOT) URINE TESTS1-6
GENETIC TESTING1-6
AHP=acute hepatic porphyria; ALA=aminolevulinic acid; PBG=porphobilinogen.
*PBG and ALA are neurotoxic intermediates that occur naturally in the heme biosynthesis pathway in the liver but can be toxic when elevated in patients with symptomatic AHP. PBG and ALA may remain elevated during recovery from an AHP attack.1,2,5,7
†Urine porphyrins can help differentiate between types of AHP.
- A PBG random (spot) urine test can measure elevated levels of PBG1
- Substantial elevation of urinary PBG is a hallmark indicator of 3 types of AHP: acute intermittent porphyria (AIP), variegate porphyria (VP), and hereditary coproporphyria (HCP)1,3
- An ALA random (spot) urine test can help confirm ALAD-deficiency porphyria (ADP)—an extremely rare type of AHP1
- PBG and ALA are neurotoxic intermediates that occur naturally in the heme biosynthesis pathway in the liver but can be toxic when elevated in patients with symptomatic AHP. PBG and ALA may remain elevated during recovery from an AHP attack
- Urine porphyrins can help differentiate among types of AHP, though it is not recommended to use urine porphyrins alone as they can be elevated for several reasons
- Genetic testing to determine AHP type can be performed at any time, regardless of the presence of symptoms at that time
- While a random (spot) urine test is the most common method to help diagnose AHP, a genetic test can be useful for at-risk family screening. However, not everyone with a mutation for AHP will have symptomatic disease
Testing for AHP: CPT Codes and Laboratory Results1,5,8
Random (spot) Urine Test
CPT Code
Laboratory Values by Acute Hepatic Porphyria (AHPs) Subtype
AIP
HCP
VP
ADP
Porphobilinogen (PBG)
84110
Increased
Increased
Increased
Increased
Delta-Aminolevulinic Acid (ALA)
82135
Increased
Increased
Increased
Increased
Porphyrins
84120
Increased URP
Increased COPRO
Increased COPRO
Increased COPRO
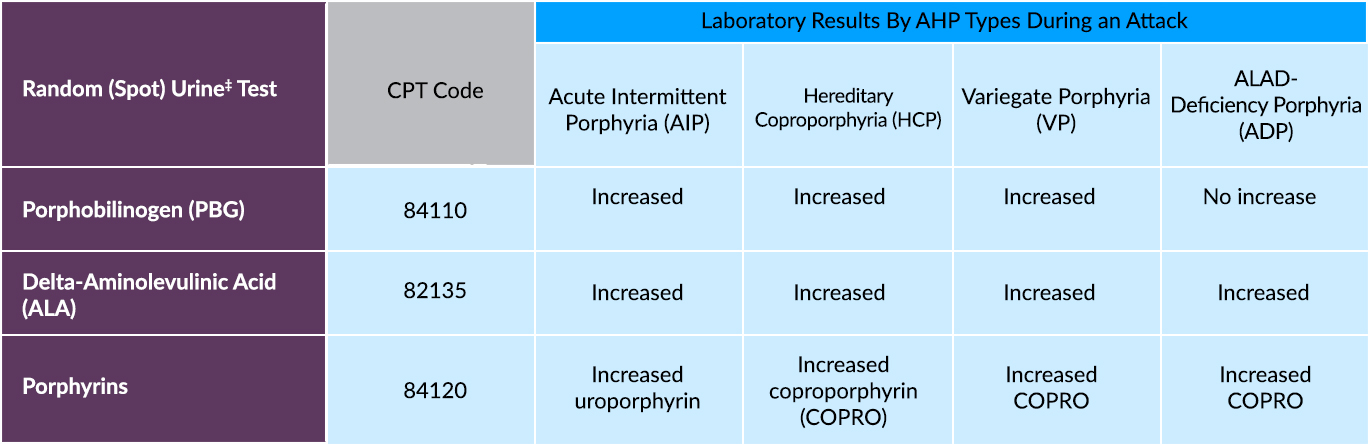
‡Tests are more accurate when normalized per gram of urine creatinine and when sample is collected during acute episodes. 24-hour urine collection is generally not required. Additional testing (genetic or biochemical) may be required to differentiate AHP type (AIP, HCP, VP, or ADP).
- Specimen requirements are lab-specific
- During acute episodes, PBG is substantially elevated9
- It is not recommended to use urine porphyrins alone to diagnose AHP, as they can be elevated for several reasons9
The optimal time to test urine is during or shortly after an attack when ALA and PBG levels have spiked, because levels may fall when symptoms resolve.
When Testing for AHP, Also Consider the Following:
- Ordering lab tests for urine porphyrins does not include assessment of ALA/PBG or their corresponding levels9
- ALA levels are substantially elevated during attacks for all 4 types of AHP, while PBG is substantially elevated for only the 3 most common forms (excluding ADP), but both ALA and PBG levels may decrease between attacks9§
- It is recommended to normalize test results to urine creatinine to avoid false negatives, especially in diluted urine samples3
- Urine color is normal, but can turn dark red or purple when exposed to a particular wavelength of light in a spectrum emission test4,6
- Samples should typically be light-protected and frozen or refrigerated based on laboratory specifications. Short delays in refrigeration/freezing should not cause false negative results. For the most up-to-date testing and protocol information, please refer to your local laboratories for specific requirements and guidance3,10
- Once a diagnosis‖ of AHP is confirmed, gene sequencing can be used to identify the mutation and the type of AHP. Consider genetic testing of family members of patients diagnosed with AHP to identify individuals who may also carry an AHP gene mutation and potentially have elevated toxic levels of ALA and PBG3
§ALA and PBG are porphyrin precursors that occur naturally in the heme biosynthesis pathway in the liver but reach toxic levels in patients with symptomatic AHP.2,5
‖Diagnosis is based on clinical judgment.
One Genetic Testing Option

Alnylam sponsors genetic testing and counseling.
The Alnylam Act® offers third-party genetic testing and counseling for patients who may have acute hepatic porphyria at no charge to patients, physicians, and payers.
The Alnylam Act® program was created to provide access to genetic testing and counseling to patients as a way to help people make more informed decisions about their health.
- While Alnylam provides financial support for this program, tests and services are performed by independent third parties
- Healthcare professionals must confirm that patients meet certain criteria to use the program
- Alnylam receives de-identified patient data from this program, but at no time does Alnylam receive patient-identifiable information. Alnylam may use healthcare professional contact information for research purposes
- Both genetic testing and genetic counseling are available in the US and Canada
- Healthcare professionals or patients who use this program have no obligation to recommend, purchase, order, prescribe, promote, administer, use, or support any Alnylam product
- No patients, healthcare professionals, or payers, including government payers, are billed for this program
References: 1. Anderson KE, Bloomer JR, Bonkovsky HL, et al. Ann Intern Med. 2005;142(6):439-450. 2. Bissell DM, Anderson KE, Bonkovsky HL. N Engl J Med. 2017;377(9):862-872. 3. Balwani M, Wang B, Anderson KE, et al; Porphyrias Consortium of the Rare Diseases Clinical Research Network. Hepatology. 2017;66(4):1314-1322. 4. Ventura P, Cappellini MD, Biolcati G, Guida CC, Rocchi E; Gruppo Italiano Porfiria (GrIP). Eur J Intern Med. 2014;25(6):497-505. 5. Pischik E, Kauppinen R. Appl Clin Genet. 2015;8:201-214. 6. Ramanujam V-MS, Anderson KE. Curr Protoc Hum Genet. 2015;86:17.20.1-17.20.26. 7. Gouya L, Bloomer JR, Balwani M, et al. Presented at: 2017 International Congress on Porphyrins and Porphyrias; June 26, 2017; Bordeaux, France. 8. Whatley SD, Mason NG, Woolf JR, Newcombe RG, Elder GH, Badminton MN. Clin Chem. 2009;55(7):1406-1414. 9. Bissell DM, Wang B. J Clin Transl Hepatol. 2015;3(1):17-26. 10. Woolf J, Marsden JT, Degg T, et al. Ann Clin Biochem. 2017;54(2):188-198.